The Digital Twin for Regulated Lab Operations
Xenox gives regulated laboratories a controlled operational layer for instruments, calibration, qualification, work orders, evidence, audit trails, and readiness.
Built for pharmaceutical QC and other regulated environments, Xenox helps teams see what is current, what needs attention, what has changed, and what evidence is available — without relying on disconnected spreadsheets, binders, and manual follow-up.
Xenox is the operational control layer for the modern regulated lab.
One Platform for Lab Operations, Evidence, and Oversight
Xenox creates a digital twin of the regulated lab by connecting core operational records with the workflows and evidence that support them.
From instrument registry and IQ/OQ/PQ to calibration, work orders, method records, audit history, and controlled AI assistance, Xenox provides a clearer view of lab readiness in one structured environment.
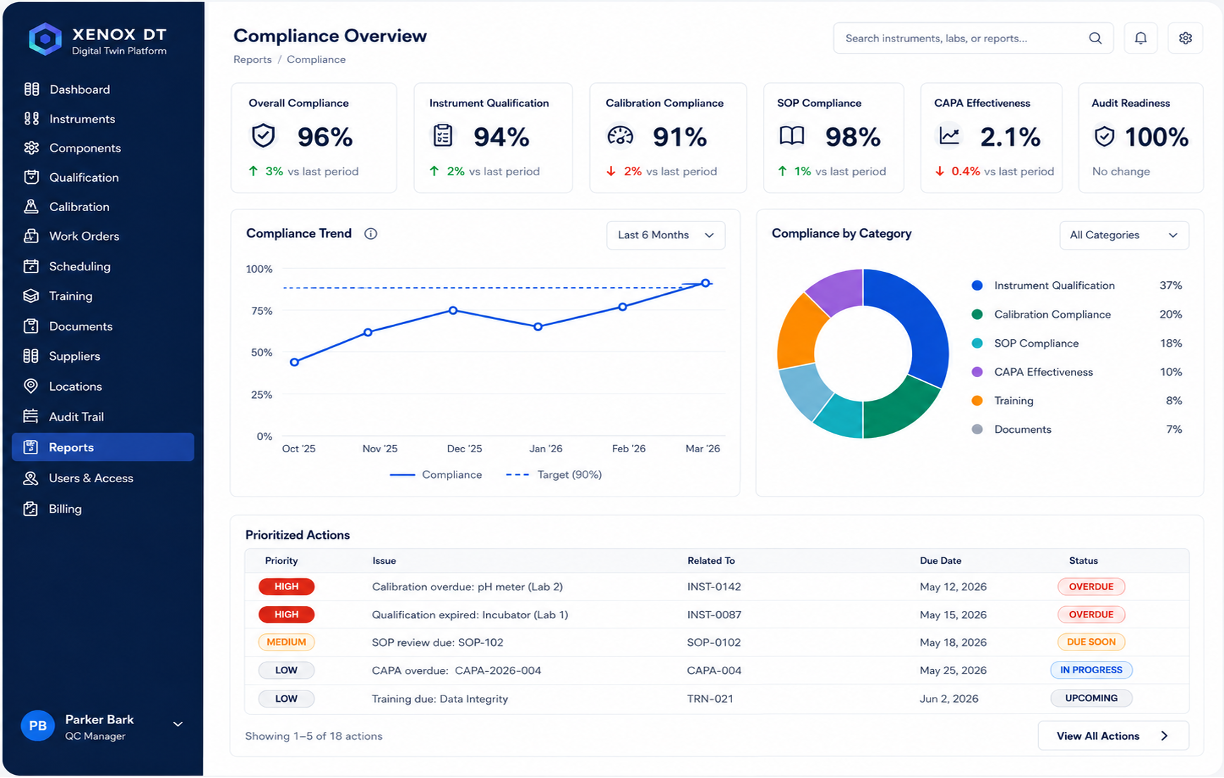
A Clearer way to Manage Regulated Lab Operations
Regulated lab operations are often fragmented across LIMS, calibration systems, document repositories, spreadsheets, binders, email, and local knowledge.
Xenox is designed to reduce that fragmentation. It helps quality, operations, and lab teams understand the status of the lab, identify gaps earlier, and prepare for inspections with greater confidence.
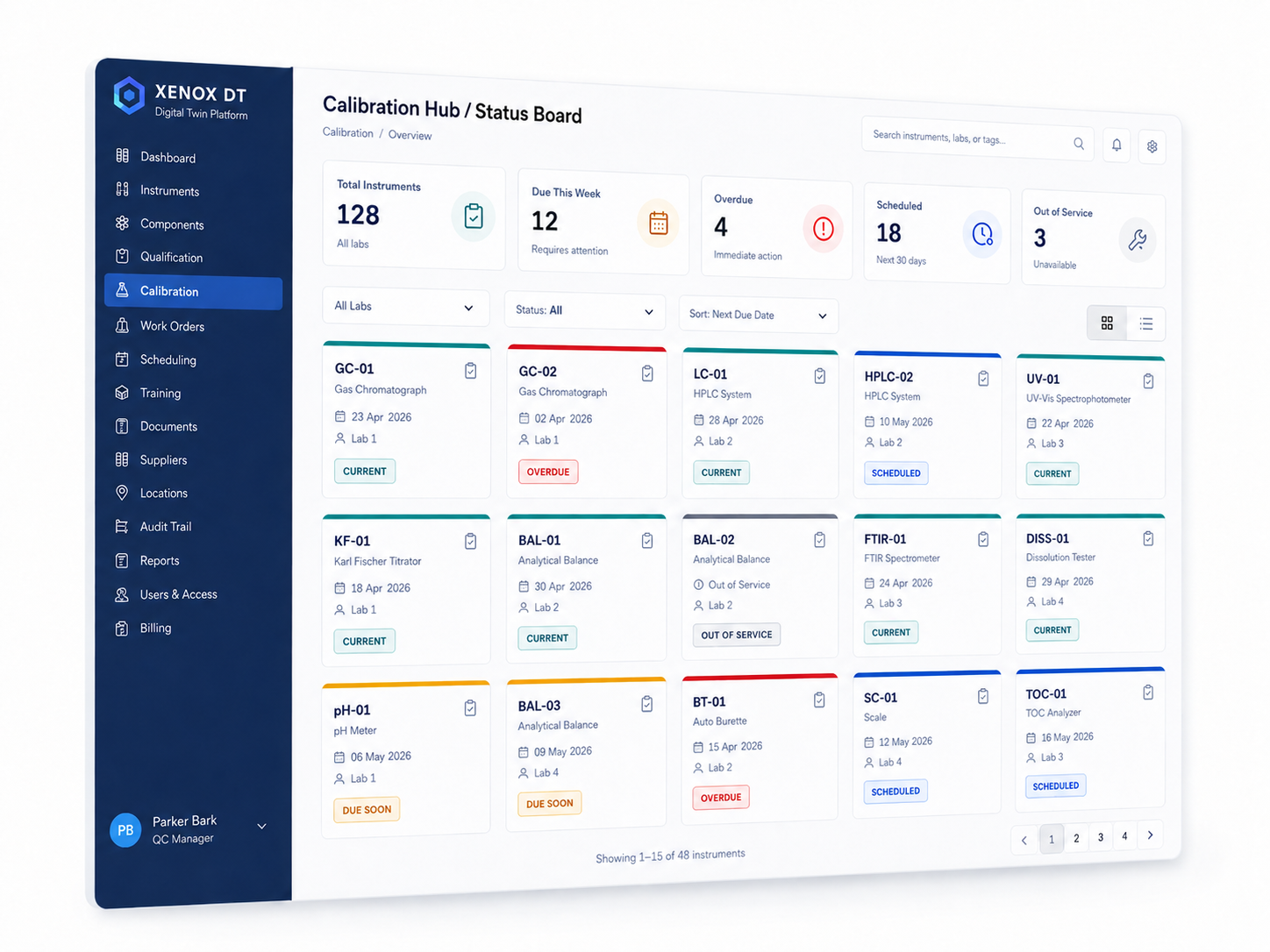
Built around the way regulated labs actually work
Xenox has been built by a highly experienced team with deep roots in LIMS, instrument management, pharmaceutical QC, cybersecurity, and enterprise software.
The platform reflects decades of experience designing and supporting systems for regulated scientific environments, combined with modern software architecture, automation, and carefully controlled use of AI.
Xenox is designed around the way labs actually operate: instruments, components, methods, documents, suppliers, locations, work orders, controlled actions, approvals, and evidence all connected in a single operational view.
